Defining Heterogeneity of Stress-induced Beta Cell Senescence Programs in Type 1 Diabetes
Contact PI: Feyza Engin, PhD, University of Wisconsin-Madison (U01 DK143458)
Sangeeta Dhawan, PhD, MPI, City of Hope
Matthew Wortham, PhD, MPI, University of Colorado Anschutz Medical Campus
Julia Panzer, PhD, Co-Investigator, City of Hope
Start Date: April 10, 2026
Abstract
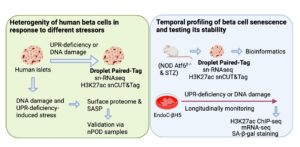
Type 1 diabetes (T1D) results from insulin insufficiency owing to near complete destruction of insulin- producing pancreatic β-cells by an autoimmune process. Over the last decade, the active participation of pancreatic β-cells in their own autoimmune destruction and the impact of aberrant stress responses to T1D disease progression have gained considerable attention. Pharmacologic agents targeting β-cell stress responses have shown promise in clinical trials, underscoring the potential for targeting β-cell stress in diabetes. Senescence is a β-cell stress adaptation program that has recently emerged as a key contributor to T1D pathogenesis. Senescence involves a cell-cycle arrest that prevents the replication of damaged cells and activation of a secretory program that promotes their clearance by the immune system. However, failure of immune surveillance leads to the accumulation of senescent cells, leading to chronic inflammation and tissue dysfunction. Our work has identified context-dependent heterogeneity of β-cell senescence, with different cellular stressors initiating distinct senescence programs with divergent effects on T1D progression. Senescent β-cells that normally accumulate in the non-obese diabetic (NOD) mouse model of T1D promote islet inflammation and disease progression. In contrast, NOD β-cells deficient for the unfolded protein response (UPR) exhibit an alternative senescence program marked by a secretome that promotes immune clearance and is protective against diabetes. Importantly, we have shown that β-cells exhibiting features of both senescence programs accumulate during T1D pathogenesis in humans. These intriguing findings highlight a serious knowledge gap regarding the nature and extent of β-cell senescence heterogeneity. As our discoveries show that distinct subsets of protective and pathogenic senescent β-cells exist in T1D, there is an urgent need to define the compendium of β-cell senescence programs and identify their underlying causes.
We hypothesize that β-cell senescence exhibits molecular and phenotypic heterogeneity in a context-dependent manner based on the nature of the initiating stressor and subsequent temporal progression. In this proposal we will (1) define the molecular and epigenetic heterogeneity of senescent β-cells in primary human islets, and (2) track the temporal progression and stability of each senescence program. We will use cutting-edge techniques including single cell multiomics as well as proteomics of cell surfaces and secretomes. Leveraging the comprehensive molecular maps of β-cell senescence heterogeneity generated in this proposal, we will mine publicly available datasets of islets from T1D and autoantibody-positive samples to define the extent of β-cell senescence heterogeneity during the pathogenesis of human diabetes. Overall, these studies will support the development of targeted interventions to maximize the beneficial effects of β-cell senescence programs while mitigating the pathological effects of maladaptive β-cell senescence programs in T1D.